- Cancer Helpline
- +91 8069420405
T cells are part of the body's immune system that helps us fight various infections and cancers. Chimeric Antigen Receptor T (CAR T) cells are genetically engineered T cells. CAR T cell Therapy harnesses the power of modified T cells to seek out and eliminate cancer cells, enhancing the body's natural defenses against the disease. This modern form of Immunotherapy offers promise in the fight against various cancers, including lymphomas, leukemias, and certain recurrent blood cancers. CAR T cell therapy is approved by United States Food and Drug Administration (USFDA) for hematological malignancies. CAR T cells identify and attack cancer cells in a patient body to stop the cancer from growing in the long term.
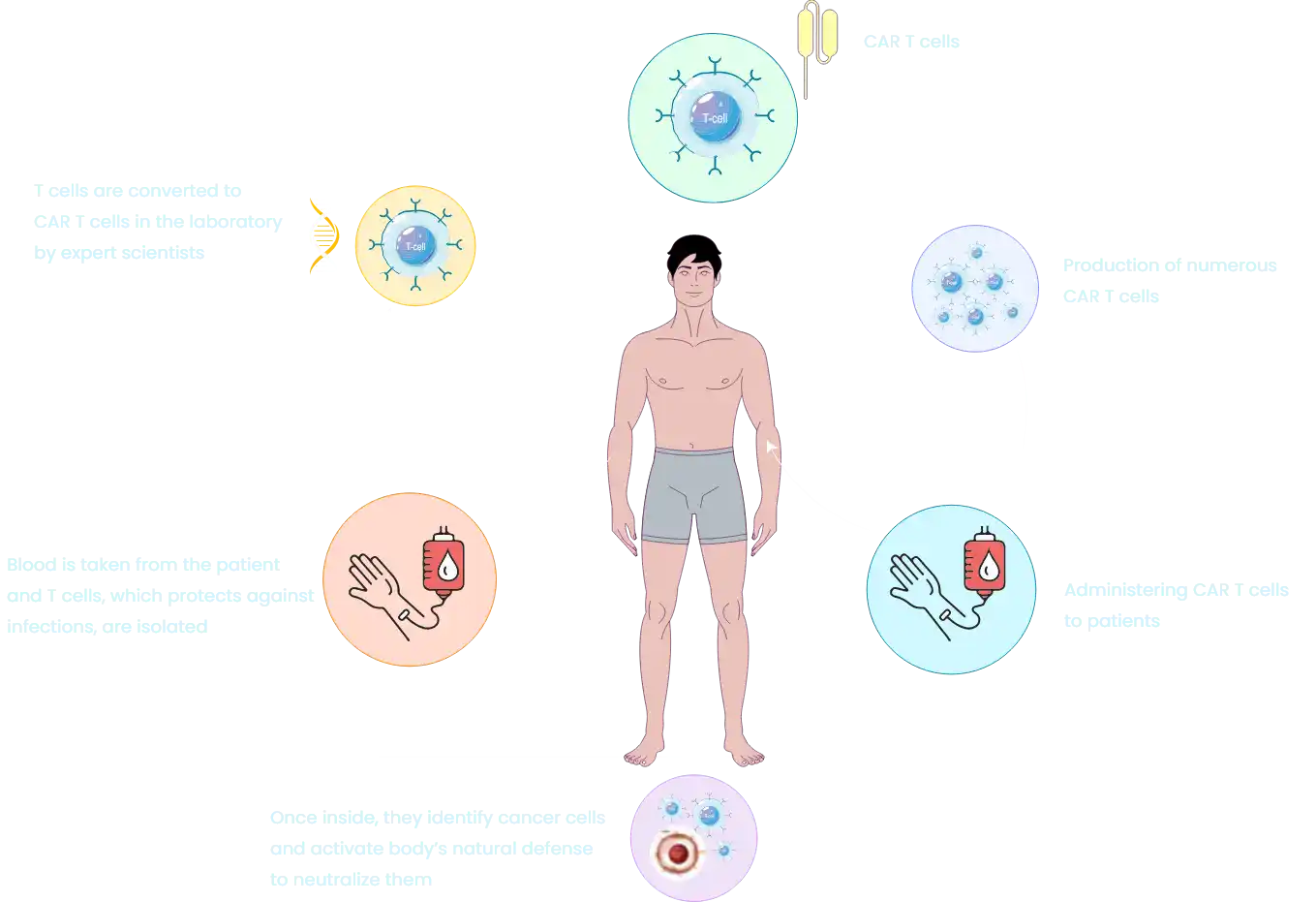
Like blood donation, blood is collected from patients to separate T cells. These T cells are then modified in the laboratory under expert supervision to express the chimeric antigen receptor. Before CAR T cell administration, patients need to undergo chemotherapy to prepare their bodies for CAR T cell therapy. Once infused, CAR T cells are anticipated to recognize and neutralize resistant cancer cells by activating the body’s immune system.Figure 1 discusses the production and function of CAR T cells.

Since 2017, the FDA has approved six CAR T-cell therapies for treating blood cancers like lymphomas, certain types of leukemia, and most recently, multiple myeloma.
One potential approach is to use CAR T-cell therapy after the initial treatment to strengthen its impact.
It's typically given after multiple rounds of treatment. CAR T cell therapy showed impressive response rates and the chance for long-lasting remission, even after previous treatments have failed.
Patients eligible for CAR T-cell therapy must meet FDA criteria and be in good overall health to handle the waiting period and potential side effects. Their T-cell health must also be sufficient for successful treatment. Seeking guidance from an oncologist is crucial to determining eligibility
With proper care and observation, patients typically recover well within 2-3 weeks.
Offers better outcomes compared to traditional cancer treatments, potentially leading to longer-term remission and improved quality of life
Demonstrated remission in patients who had previously undergone multiple unsuccessful cancer treatments
Quick intervention time and single infusion of CAR T cells
Shorter
treatment time
Advantageous in targeting and destroying cancer cells during disease relapse
Consultation: You will have detailed discussions with your oncologist to ensure you are a suitable candidate for therapy.
Pre-Treatment Tests: Once your eligibility has been determined, several tests may be conducted to evaluate your health status.
Apheresis: This process will take a few hours and is generally performed in an outpatient department setting.
Infusion: After the laboratory work, the infusion of CAR T cells is carried out in a controlled environment. This often requires a brief hospital stay for close monitoring of the patient.
Monitoring: Once the cells have been infused, you will be closely monitored for any immediate side effects, such as fever or chills.
Follow-Up Care: Regular follow-up appointments will be scheduled to assess the effectiveness of the therapy and manage side effects if any.

While CAR T cell therapy represents a significant advancement in cancer treatment, it's natural to have concerns about any medical procedure. Rest assured that this therapy has undergone rigorous testing and has shown promising results for many patients with otherwise difficult-to-treat cancers. Your doctors will provide continuous support throughout your treatment journey, ensuring you receive the best possible care at every step. Consult with your doctor to explore whether this ground-breaking treatment is right for you. Stay informed, stay hopeful, and let’s fight cancer together with cutting-edge therapies like CAR-T cell therapy.